Health
Remdesivir, which WHO found has little or no effect in reducing coronavirus deaths, shows mixed results in Nepal
Nepal Health Research Council observes that the antiviral medication cut hospital stay of serious patients, lessened symptoms and also reduced deaths.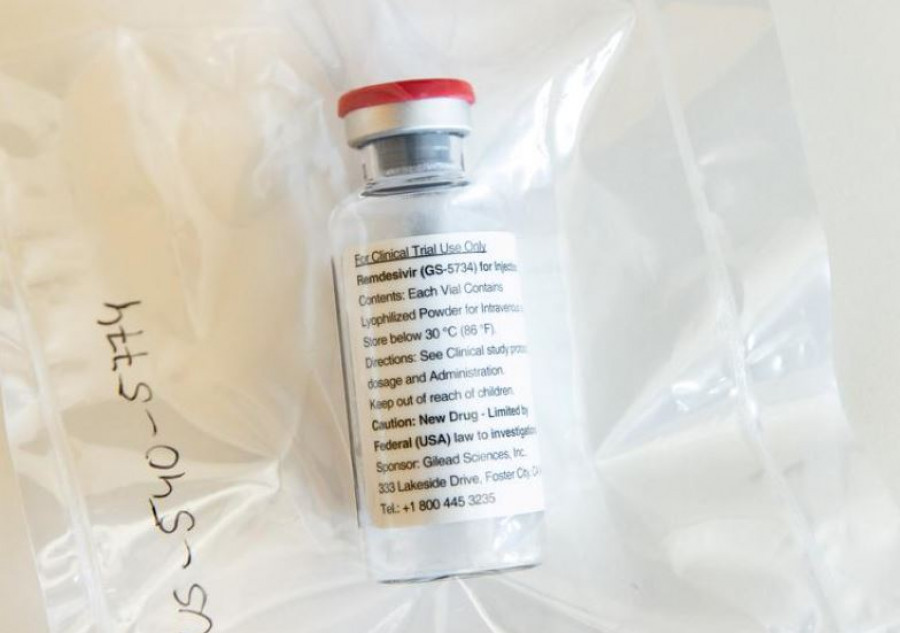
Arjun Poudel
Contrary to the findings of Solidarity, an international clinical trial carried out by the World Health Organization, antiviral medication remdesivir showed mixed results in Nepal.
According to the Nepal Health Research Council, which has been overseeing the effects of remdesivir on patients, the antiviral medication was found to be effective in the treatment of serious Covid-19 cases.
“Yes, our study shows the antiviral medicine [remdesivir] reduced hospital stay, lessened symptoms and reduced deaths also,” Dr Janak Koirala, clinical trial consultant at the council, told the Post.
The study was performed on 1,200 moderately and seriously ill patients, who were given oxygen therapy due to low oxygen saturation, admitted to intensive care units or placed on ventilator support.
Solidarity, one of largest international randomised trials for Covid-19, involved 12,000 patients admitted in 500 hospital sites of over 30 countries, according to the UN health agency.
Apart from remdesivir, hydroxychloroquine, lopinavir/ritonavir and interferon were also used in the WHO study. The trial evaluated effects of the drugs on three important outcomes in Covid-19 patients: mortality, need for assisted ventilation and duration of hospital stay.
“It found that all four treatments [remdesivir, hydroxychloroquine, lopinavir/ritonavir and interferon] had little or no effect on overall mortality, initiation of ventilation and duration of hospital stay in hospitalised patients,” WHO said in its interim results published on October 15.
When asked about the WHO’s interim report, Dr Pradip Gyanwali, executive chief of Nepal Health Research Council, said that the WHO’s interim report is yet to be peer- reviewed, which means the report should be scrutinised by experts from the same field to ensure its scientific quality.
“A final report with data is yet to be completed but our preliminary reports show mixed results,” Dr Gyanwali told the Post. “We will conclude this study within a week and publish the results.”
The council said that a preliminary report was prepared on the basis of data of 345 patients involved in the study.
The Department of Drug Administration, the national regulator of the drug market, in September had authorised the import and usage of remdesivir for treatment of Covid-19 patients, as a study drug and delegated authority to the council to oversee its use.
After the report of the council is unveiled, the administration could permit traders to import it like other drugs and sell it in the market.
On October 22, the US Food and Drug Administration approved remdesivir for use in adults and pediatric patients 12 years of age and older and weighting at least 40 kilograms for the treatment of Covid-19 requiring hospitalisation.
The UN health agency, however, cautioned against physicians and medical associations recommending or administering unproven treatments to patients of Covid-19 or people self-medicating with them.
With exponential rise in new infections, a number of symptomatic and serious cases have also been rising throughout the country. Hospitals designated for Covid-19 treatment have been overwhelmed and thousands of infected patients have been deprived of treatment.
As of Saturday, 170,743 people across Nepal have been infected, with 937 deaths. In the last 24 hours, 2,508 people tested positive including 1,357 in Kathmandu Valley. The Health Ministry said there were 38,584 active cases on Saturday.




 26.18°C Kathmandu
26.18°C Kathmandu