Health
Plans afoot to buy additional doses of Covid-19 vaccine, officials say
Government to procure four million doses within a month, but some basic preparations like making guidelines and training health workers are yet to be completed.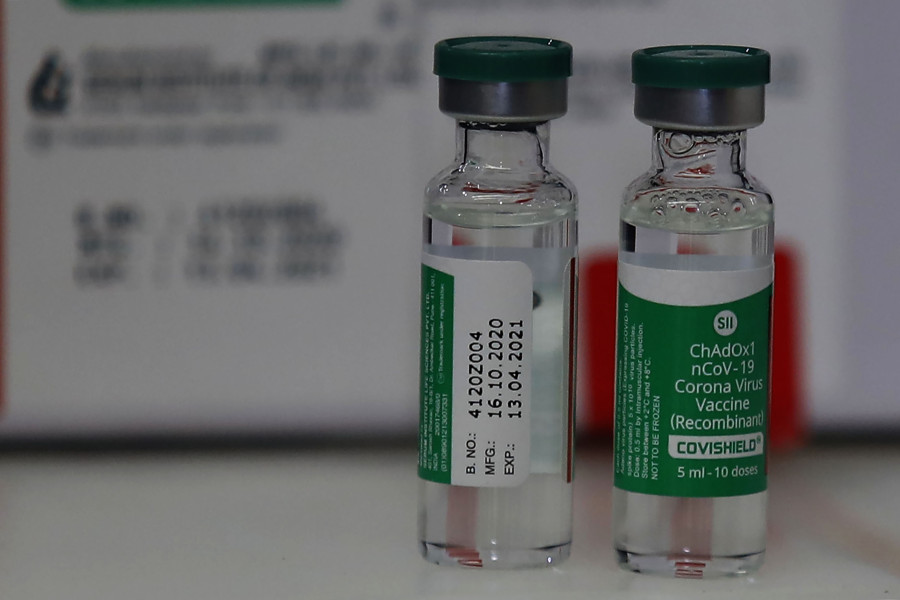
Arjun Poudel
After receiving one million doses of Covid-19 vaccine under grant assistance from India, the government has said it will procure additional four million doses within a month.
At least two officials at the Ministry of Health told the Post that Health Minister
Hridayesh Tripathi has conveyed to those within his orbit that four million doses will arrive in Nepal within a month.
“Preparations are underway to procure additional doses, either at subsidised rates or under government-to-government deal or through direct procurement deals with India’s vaccine manufacturing companies,” said an official requesting anonymity because the discussion is at a preliminary phase.
India has provided one million doses of Covishield, a Covid-19 vaccine produced by the Serum Institute of India. The vaccine was developed by the University of Oxford and pharmaceutical giant AstraZeneca.
Officials say since Covishield is Nepal’s preferred choice of vaccine—the existing storage and transportation infrastructure used in the country to immunise children can be utilised—the government is likely to procure more of the same vaccine.
Covishield vaccine can be stored in temperatures between 2 to 8 degrees Celsius.
“As per my understanding, the current focus is on importing Covishield manufactured by the Serum Institute of India,” Dr Shyam Raj Upreti, coordinator of Covid-19 Vaccine Advisory Committee, told the Post.
The government has decided to procure vaccines that have received emergency approval and administer them to people free of charge on the basis of priority.
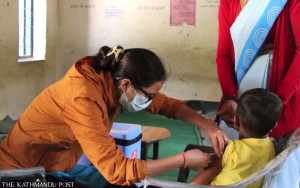
The government plans to inoculate around six million people (20 percent of population) in the first phase and frontline workers—health workers, supporting staff, sanitation workers, ambulance drivers, female community health volunteers, security personnel deployed on the frontline—have been prioritised.
The country needs to inoculate 72 percent of its 30 million people, as children up to 14 years of age, who constitute 28 percent of the population, can’t be immunised because vaccines haven’t been tested on them. Covishield, however, can be administered to only those who are above 18 years of age.
According to the Serum Institute of India’s fact sheet, the vaccine has been given a restricted use licence for emergency situations.
“It does not have a marketing authorisation, however, this approval for the restricted use in emergency situations grants permission for the vaccine to be used for active immunisation of individuals aged 18 years and older for the prevention of Covid-19,” says the fact sheet.
The company has said this vaccine will be closely monitored to allow quick identification of new safety information.
The vaccine vials that arrived on Thursday, according to officials, will be delivered to provincial vaccine storage facilities, which will later supply them to district hospitals maintaining the cold chain. The process is likely to be completed within two to three days, according to officials. A decision on vaccine rollout has to be taken by the Cabinet, they said.
“It’s a concern for all of us, as we have been waiting for the vaccines to arrive, a political decision will be made regarding its rollout,” Dr Samir Kumar Adhikari, joint spokesperson for the Health Ministry, told the Post.
Officials believe there won’t be a long gap between the first and second phases if the additional four million doses arrive within a month.
The government is planning to launch the vaccination drive simultaneously at all district hospitals.
Each team will administer the vaccine to 100 people in a day and plans are afoot to run immunisation stations at about five hospitals in densely populated districts such as Kathmandu in the first phase.
“Elderly people above 55 years will get priority when we launch the next phase of inoculation,” said Adhikari.
The government has estimated that around Rs48 billion would be required to inoculate 52 percent of the population, as vaccines for 20 percent of the population will be provided by the World Health Organisation under its COVAX programme. The government doesn’t need to spend money for the vaccines to be received under the COVAX facility. Officials say the World Health Organisation has assured that vaccines to inncoulcate 3 percent of the population will be provided to Nepal by April.
The vaccines provided by India, however, will be sufficient to inoculate all on the first priority list prepared by the Ministry of Health and Population.
There are over 911,000 frontline workers, including health workers, supporting hospital staff, ambulance drivers, female community health volunteers and security personnel deployed on the front lines and those involved in handling infected people and the bodies of those who die of Covid-19.
The one million doses will be sufficient for inoculating around 400,000 people, as each person needs two doses. Some doses of vaccine often go to waste due to various factors.
Officials said they are working to ensure minimum wastage as a name list of those getting the jabs has already been prepared and vaccines will be dispatched accordingly.
If additional four million doses arrive within a month, around two million people will be immunised soon.
Once rolled out, people will be inoculated within seven to ten days with the one million doses of vaccines, according to officials. But for the rollout, some preparations are yet to be made, they say.
“We have not yet printed the guidelines, which we have sent to the local level. Training for health workers has not started,” an official at the Child Health Section at the Family Welfare Division under the Department of Health Services told the Post. “In-person training is necessary for health workers serving under the provinces.”
The government also needs to work out a different plan for inoculating the population that is above 14 but under 18 years of age.
“The Oxford-AstraZeneca vaccine has not been trialled in the population under 18,” Upreti, the coordinator of Covid-19 Vaccine Advisory Committee, told the Post. “The government has to buy vaccines from other companies, which have been trialled in people under 18, if it has to inoculate people between 14 and 18 years.”
Experts say the government must not waste time in red tape and work on a war footing to secure additional doses of vaccines to protect its people from Covid-19.
According to them, the government also should not wait for the vaccines to arrive under the COVAX facility, as vulnerable groups need to be vaccinated as soon as possible.
“We can use the COVAX facility’s vaccine for people in the third or fourth phase,” Dr Bhagwan Koirala, chairman of Nepal Medical Council, the regulatory body of medical doctors, told the Post. “The government should try its best to fulfil its commitment and responsibility to save the lives of people.”
The Indian consignment of Covishield arrived in Nepal, six days after Nepal’s drug regulator granted emergency use approval for Oxford-AstraZeneca vaccine and five days after India rolled out its pan-Indian vaccination drive, the world’s largest inoculation exercise.
Nepal received its first lot of vaccines just days before it marked one year since it reported the first Covid-19 case. The first coronavirus case in Nepal was confirmed on January 24 after a man who had returned from Wuhan of China tested positive for the virus.
As of Thursday, Nepal has reported 268,646 coronavirus cases with 1,979 deaths. So far, 262,868 people have recovered, according to the Health Ministry.
Experts say the government must also launch programmes so as to make the people aware of the vaccines they are being administered and the possible side effects.
The Serum Institute of India in its fact sheet has listed the possible side effects of the vaccine based on the reports it has received after the rollout.
According to the company, very common, which may be seen in more than 1 in 10 people, side effects include tenderness, pain, warmth, redness, itching, swelling or bruising where the injection is given.
People may feel generally unwell, tired or can have chills or a feverish feeling. Nausea and joint pain or muscle ache are other possible side effects.
Other common side effects, which may be seen in up to 1 in 10 people, include a lump at the injection site, fever, being sick (vomiting), flu-like symptoms (high temperature, sore throat, runny nose, cough and chills).
Some uncommon side effects, which may be seen in up to 1 in 100 people, include feeling dizzy, decreased appetite, abdominal pain, enlarged lymph nodes, excessive sweating and itchy skin or rash.
Dr Mingmar Gyelgen Sherpa, former director general at the Department of Health Services, said authorities should not compromise on the quality while procuring vaccines and try to secure quality products as soon as possible.
“As our existing vaccine storage facility is suitable for Covishield, it will not be very difficult to get the jabs and launch the inoculation drive,” said Sherpa. “Authorities should try their best to secure additional doses as early as possible for the people in the age group facing very high risk.”




 26.18°C Kathmandu
26.18°C Kathmandu